



Staying on Treatment With INCRELEX®
INCRELEX® (mecasermin) dosing is dependent on many factors, such as tolerability and a child’s weight and height. Regular weight monitoring throughout treatment is important for appropriate dosing.
On this page

Age 10: 3’ 11”
Age 18: 4’ 10.5”
Olive, a former INCRELEX patient, at age 18
How to Inject INCRELEX®
Watch this video to find out how to prepare and inject the correct dose of INCRELEX®.
Goal of Treatment
There is a window of time when your child is able to grow. Once your child enters the teen years, the potential to grow slows – and eventually stops – when the growth plates (epiphyses) close.
What to Expect With INCRELEX®
Results
The average rate of growth (referred to as height velocity) sustained by 5 cm per year in years 2 through 6 of treatment.
Patients experienced increased rate of height growth without increasing the rate of chronological bone age. Bone age is used to categorize the degree of skeletal maturation.
*Forty-nine subjects were included in an analysis of the effects of Increlex on the way your child’s bone changes in size and shape (referred as bone age). The mean ± SD change in chronological age was 4.9 ± 3.4 years and the mean ± SD change in bone age was 5.3 ± 3.4 years.
In clinical trials, INCRELEX® improved statural growth in patients diagnosed with Severe Primary IGFD
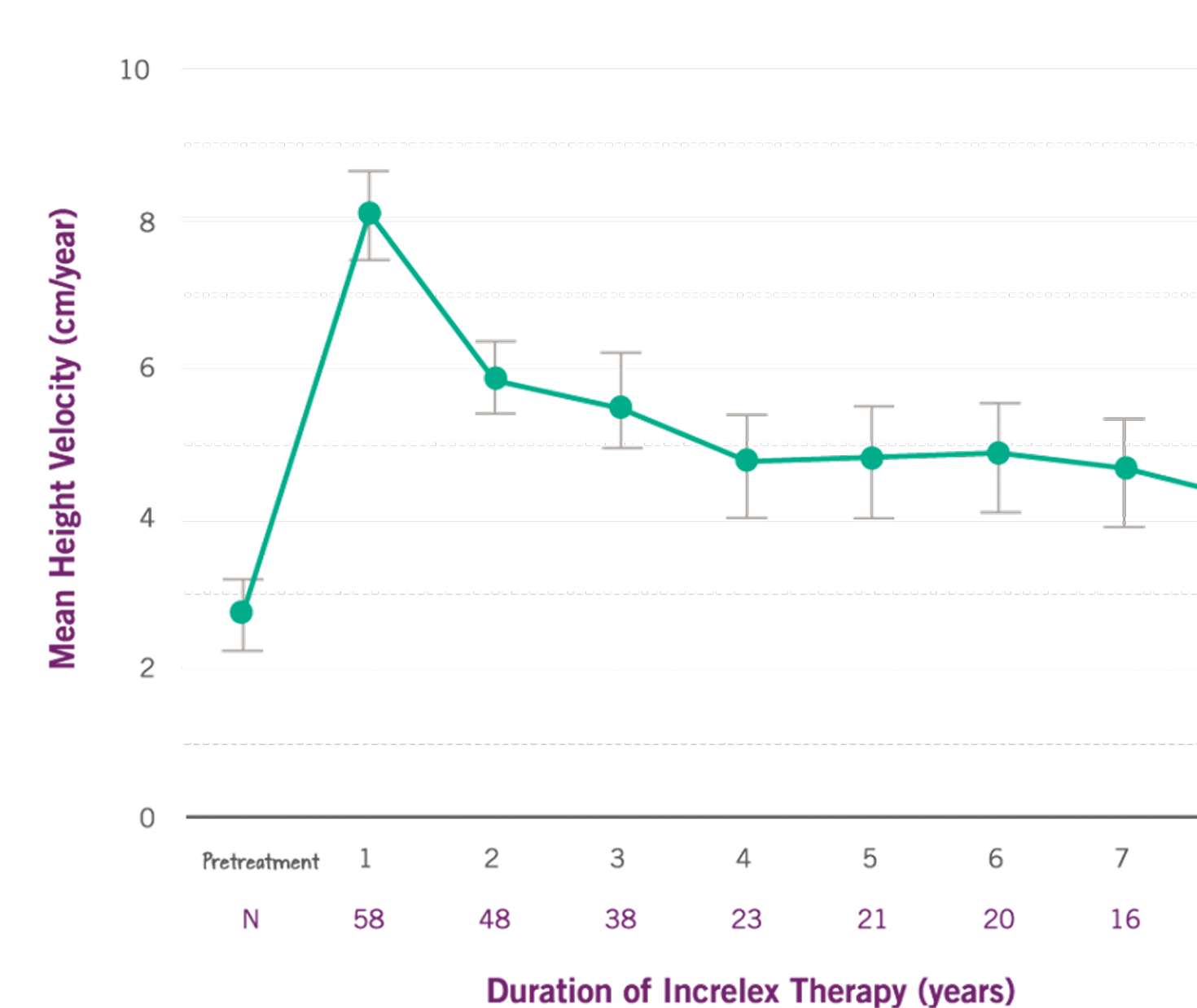
Growth Rate with INCRELEX® Over 8 Years*
What to Know About INCRELEX® Dose Adjustment
INDICATION
INCRELEX® (mecasermin) is a prescription medicine used in children 2 years and older with short stature that have severely low levels of the hormone, insulin-like growth factor-1 (IGF-1). IGF-1 is needed for normal growth.
INCRELEX is not a substitute for growth hormone. It cannot be used for children with short stature due to low levels of growth hormone, malnutrition, when their thyroid gland doesn’t make enough thyroid hormone, or when taking anti-inflammatory steroids used to manage inflammation.
IMPORTANT SAFETY INFORMATION
Always give INCRELEX exactly as your doctor directed.
Do not take INCRELEX if you are allergic to IGF-1 or any of its other ingredients. Report any allergic reactions.
INCRELEX should be injected under the skin (subcutaneous injection). It should not be injected directly into a blood vessel.
INCRELEX should not be used after growth plates close which happens during puberty.
INCRELEX should not be used in children with cancerous tumors or a history of cancer.
Hypoglycemia (low blood sugar): INCRELEX should be administered 20 minutes before or after a meal or snack and should not be administered when the meal or snack is skipped.
Checking blood glucose levels is recommended. The dose of INCRELEX may need to be adjusted until an appropriate dose is decided by your doctor.
Intracranial Hypertension: Increased pressure in your skull may occur because of cerebrospinal fluid buildup around your brain. Therefore, your doctor may require an eye examination at the start of INCRELEX treatment and periodically during the time you are taking INCRELEX.
Lymphoid Tissue Hypertrophy: Lymphoid tissue hypertrophy is a noncancerous increase in the number of immune cells called lymphocytes. Patients should have periodic examinations with your doctor to rule out potential complications.
Slipped Capital Femoral Epiphysis: Slipped capital femoral epiphysis is a bone problem where the top of the upper leg (femur) slips apart. This may lead to a serious condition where bone tissue dies due to a lack of blood supply (osteonecrosis). Get medical help for your child right away if your child develops a limp or has hip or knee pain.
Progression of Scoliosis: Your doctor will monitor you during treatment with INCRELEX if you have a history of scoliosis.
Malignant Neoplasia: There have been reports of cancerous tumors in children who received INCRELEX. It is unknown whether there is any relationship between INCRELEX therapy and new occurrence of tumors. Tumors were mostly reported in patients with rare genetic conditions of short stature associated with a higher risk of cancer, or in patients already at risk of cancer.
The tumors were seen more frequently in patients who received INCRELEX at higher than recommended doses or at doses that produced IGF-1 levels above normal for age and sex. Your doctor will carefully monitor you during your treatment with INCRELEX for development of tumors. If cancerous tumors develop, your doctor will stop your INCRELEX treatment.
Risk of Serious Adverse Reactions in Infants due to Benzyl Alcohol Preserved Solution: Serious and fatal adverse reactions can occur in neonates and infants treated with benzyl alcohol-preserved drugs. Use of INCRELEX in infants is not recommended.
The most common adverse reactions include low blood sugar, reactions at the injection site or throughout your body, and enlarged tonsils.
You are encouraged to report negative side effects of prescription drugs by contacting Eton Pharmaceuticals, Inc. at 1-855-224-0233 or the U.S. Food and Drug Administration (FDA) at www.fda.gov/safety/Medwatch or call 1-800-FDA-1088.
Please see full Prescribing Information for more information.
INDICATION
INCRELEX® (mecasermin) is a prescription medicine used in children 2 years and older with short stature that have severely low levels of the hormone, insulin-like growth factor-1 (IGF-1). IGF-1 is needed for normal growth.
INCRELEX is not a substitute for growth hormone. It cannot be used for children with short stature due to low levels of growth hormone, malnutrition, when their thyroid gland doesn’t make enough thyroid hormone, or when taking anti-inflammatory steroids used to manage inflammation.
IMPORTANT SAFETY INFORMATION
Always give INCRELEX exactly as your doctor directed.
Do not take INCRELEX if you are allergic to IGF-1 or any of its other ingredients. Report any allergic reactions.
INCRELEX should be injected under the skin (subcutaneous injection). It should not be injected directly into a blood vessel.
INCRELEX should not be used after growth plates close which happens during puberty.
INCRELEX should not be used in children with cancerous tumors or a history of cancer.
Hypoglycemia (low blood sugar): INCRELEX should be administered 20 minutes before or after a meal or snack and should not be administered when the meal or snack is skipped.
Checking blood glucose levels is recommended. The dose of INCRELEX may need to be adjusted until an appropriate dose is decided by your doctor.
Intracranial Hypertension: Increased pressure in your skull may occur because of cerebrospinal fluid buildup around your brain. Therefore, your doctor may require an eye examination at the start of INCRELEX treatment and periodically during the time you are taking INCRELEX.
Lymphoid Tissue Hypertrophy: Lymphoid tissue hypertrophy is a noncancerous increase in the number of immune cells called lymphocytes. Patients should have periodic examinations with your doctor to rule out potential complications.
Slipped Capital Femoral Epiphysis: Slipped capital femoral epiphysis is a bone problem where the top of the upper leg (femur) slips apart. This may lead to a serious condition where bone tissue dies due to a lack of blood supply (osteonecrosis). Get medical help for your child right away if your child develops a limp or has hip or knee pain.
Progression of Scoliosis: Your doctor will monitor you during treatment with INCRELEX if you have a history of scoliosis.
Malignant Neoplasia: There have been reports of cancerous tumors in children who received INCRELEX. It is unknown whether there is any relationship between INCRELEX therapy and new occurrence of tumors. Tumors were mostly reported in patients with rare genetic conditions of short stature associated with a higher risk of cancer, or in patients already at risk of cancer.
The tumors were seen more frequently in patients who received INCRELEX at higher than recommended doses or at doses that produced IGF-1 levels above normal for age and sex. Your doctor will carefully monitor you during your treatment with INCRELEX for development of tumors. If cancerous tumors develop, your doctor will stop your INCRELEX treatment.
Risk of Serious Adverse Reactions in Infants due to Benzyl Alcohol Preserved Solution: Serious and fatal adverse reactions can occur in neonates and infants treated with benzyl alcohol-preserved drugs. Use of INCRELEX in infants is not recommended.
The most common adverse reactions include low blood sugar, reactions at the injection site or throughout your body, and enlarged tonsils.
You are encouraged to report negative side effects of prescription drugs by contacting Eton Pharmaceuticals, Inc. at 1-855-224-0233 or the U.S. Food and Drug Administration (FDA) at www.fda.gov/safety/Medwatch or call 1-800-FDA-1088.
Please see full Prescribing Information for more information.
